
For progression-free survival, hazard ratios were 0.66, 0.75, and 0.82, respectively. The primary analysis revealed an overall survival hazard ratio of 0.73 among patients with a CPS of at least 10, 0.68 for those with a CPS score of at least 1, and 0.89 in the intent-to-treat population. Participants were randomly assigned to receive nab-paclitacel, paclitaxel, or gemcitabine and carboplatin, plus either pembrolizumab or placebo for up to 35 doses, until intolerable toxicity or progressive disease. The investigators focused on 847 patients with previously untreated, locally recurrent, inoperable or metastatic triple-negative breast cancer. 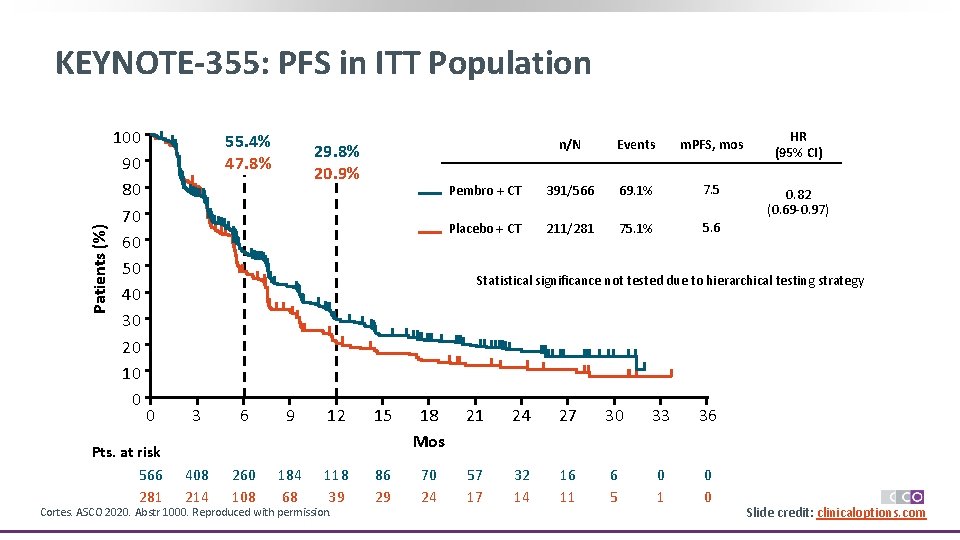
Presented during the 2021 San Antonio Breast Cancer Symposium (SABCS Abstract GS1-02), their final results demonstrated that the addition of pembrolizumab yielded significant survival improvements over placebo and suggest that a combined positive score (CPS) of at least 10 is a “reasonable” cutoff to determine expected treatment benefit. 
The phase III KEYNOTE-355 study, which evaluated pembrolizumab plus chemotherapy versus chemotherapy alone in patients with previously untreated, locally recurrent, inoperable or metastatic triple-negative breast cancer, was conducted by Javier Cortes, MD, PhD, of the International Breast Cancer Center, Madrid, and colleagues. Carter, BS Posted: Tuesday, December 14, 2021 SABCS 2021: KEYNOTE-355 Final Results of Pembrolizumab Plus Chemotherapy in Triple-Negative Breast Cancerīy: Vanessa A.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
